 
The Bohr atomic model theory states that atoms are composed of a nucleus, which consists of one or more protons and neutrons that are held together by nuclear forces. The electrons sit in energy levels around the nucleus, with the lowest possible energy level being electron number one and the highest being electron number eight. Atoms consist of a central nucleus, surrounded by electrons in orbital shells. 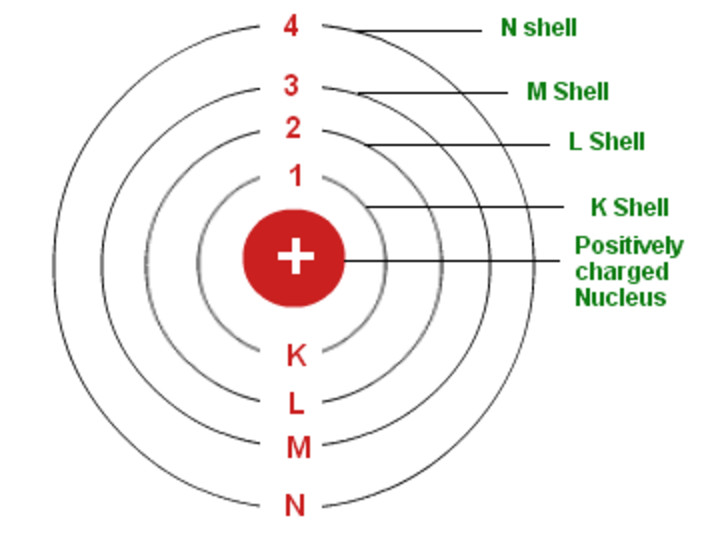
In his model, Bohr postulated that electrons were placed in orbits that are referred to as orbitals. Niels Bohr’s atomic model was created based on previous research by Rutherford, Rutherford’s gold foil experiment, and Ernest Rutherford’s model of the atom. It was a postulation of Bohr that the electrons rotated in a circular orbit around the nucleus of the atom. The Niels Bohr Atomic Model theory is a model that was introduced by Niels Bohr in 1913 to describe the atom. What Was Niels Bohr Experiment? What Did Niels Bohr Discover? The atomic model is a theory that holds that the atoms in an element are different from one another and contain protons, electrons, and neutrons.īohr’s theory involved having electrons orbiting the nucleus in orbits and when they have near the nucleus and have an orbit with the same angular momentum, they could not change orbits without the release of some form of energy. Niels Bohr is a well-known Danish physicist that spent the majority of his life studying the atomic model. Niels Bohr Atomic Model Theory Experiment Niels Bohr Education & Life
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
